Introduction
Cooking is a traditional processing technology widely used to ensure safety and enhance palatability attributes of meats (Tornberg, 2005; Sarah et al., 2014). Muscle proteins undergo structural modifications during thermal processing (Tian et al., 2016), and the denaturation of myoglobin (Mb) results in the dull-brown color of cooked meats (King and Whyte, 2006). The cooking process denatures Mb resulting in a dull-brown appearance in the interior of the product. Therefore, biochemistry of cooked color differs from the color phenomena in fresh meats, wherein the heme protein is in its native state. Nonetheless, the relationship is strong between the chemistry of Mb in raw and cooked meats (King and Whyte, 2006; Suman and Joseph, 2013; Suman et al., 2016). Mb denaturation starts at 55°C and is completed at 75°C–80°C (Hunt et al., 1999; King and Whyte, 2006); therefore, cooked color depends on the endpoint temperature (Mancini and Hunt, 2005; Suman et al., 2016).
Sarcoplasmic proteome comprises water-soluble proteins (including enzymes and Mb) and governs biochemical pathways influencing meat color (Renerre et al., 1996). Proteomic techniques have been applied extensively to explain the role of sarcoplasmic proteome components in color biochemistry of fresh beef (Joseph et al., 2012; Canto et al., 2015; Gagaoua et al., 2015; Nair et al., 2016; Gagaoua et al., 2017; Gagaoua et al., 2018; Mahmood et al., 2018; Nair et al., 2018). However, limited investigations have utilized proteomic tools to examine biochemistry of cooked meat color. Sarah et al. (2014) studied the influence of heat treatments (boiling at 100°C and autoclaving at 121°C) on goat longissimus thoracis muscle proteins and documented a high heat stability of Mb compared with other muscle proteins. Li et al. (2018) compared the influence of cooking method and temperature (water bath at 78°C; boiling at 100°C; sterilizing at 121°C; frying and baking at 200°C) on thermal stability of beef proteins and reported Mb among the thermally stable proteins. Tian et al. (2016) investigated the influence of different cooking methods (water bath, short-time ohmic cooking, and long-time ohmic cooking) on color and proteome profile of beef longissimus cooked to 72°C and observed greater redness (a* value) and overabundance of chaperones and glycolytic enzymes in meat subjected to short-time ohmic cooking than in their counterparts cooked with other techniques.
Nonetheless, the relationship between sarcoplasmic proteome and color attributes in beef cooked to different internal endpoint temperatures has not been examined yet. Therefore, the objective of the present study was to examine the changes in sarcoplasmic proteome and their influence on the internal color of beef longissimus lumborum (LL) steaks cooked to different endpoint temperatures.
Material and Methods
The beef strip loins were purchased from a commercial packing plant. Therefore, Institutional Animal Care and Use Committee approval was not obtained.
Beef muscle fabrication and evaluation of fresh surface color
Nine (n = 9) USDA Choice beef strip loins (14 d post mortem; IMPS #180; NAMP, 2010) were obtained from a commercial packing plant and were transported to the USDA-inspected meat laboratory at the University of Kentucky (Lexington, KY). Two 2.54-cm-thick LL steaks were fabricated from the center using a knife and were randomly assigned for cooking at 60°C (C-60) and 71°C (C-71) internal endpoint temperatures.
Immediately after fabrication, the steaks were allowed to bloom at 2°C for 2 h before instrumental evaluation of fresh color. After blooming, the reflectance spectra (from 400 to 700 nm) and CIE L*, a*, and b* values were measured at 3 random locations on the bloomed surface using a HunterLab LabScan XE colorimeter (Hunter Associates Laboratory, Reston, VA) with 2.54-cm-diameter aperture, illuminant A, and 10° standard observer (AMSA, 2012). Additionally, the ratio of reflectance at 630 and 580 nm (R630/580) was used to estimate the relative amount of metmyoglobin (AMSA, 2012).
Cooking and cooked color evaluation
Following the raw surface color analyses, steaks were cooked to an internal endpoint temperature of either 60°C or 71°C in a George Foreman clam-shell grill (Salton Inc., Columbia, MO) with a surface temperature of 180°C (Suman et al., 2009; Suman et al., 2010). The internal temperature of the steaks was continuously monitored using a handheld probe thermometer (AccuTuff 340, Atkins, Gainesville, FL) inserted into the geometric center. Steaks were removed from the grill after reaching the desired internal temperature and were allowed to cool for 5 min.
Five minutes after removal from the grill, cooked steaks were cut parallel to the grilled surface, and the internal color was evaluated. Instrumental color was measured using a HunterLab LabScan XE colorimeter as previously described. Immediately after evaluation of cooked color, an approximately 10-g sample was collected from the interior core of cooked steaks using a knife and was immediately vacuum packaged and frozen at −80°C for sarcoplasmic proteome analyses.
Isolation of sarcoplasmic proteome
The samples collected from cooked steaks were used for sarcoplasmic proteome isolation. Frozen tissue (10 g) was homogenized in 25-mL ice-cold extraction buffer (40 mM Tris, 2 mM ethylenediaminetetraacetic acid, pH 8.0) using a blender (Model No. 51BL32; Waring Commercial, Torrington, CT), and the homogenate was centrifuged at 10,000g for 15 min at 4°C. The supernatant consisting of the sarcoplasmic proteome extract was filtered (Whatman No. 1 filter paper) and utilized (Joseph et al., 2012; Nair et al., 2018).
Two-dimensional electrophoresis
The protein concentration of the sarcoplasmic proteome extract was determined by employing Bradford assay (Bradford, 1976) using the Bio-Rad protein assay kit (Bio-Rad, Hercules, CA). The sarcoplasmic proteome (600 μg) was mixed with DeStreak rehydration solution (GE Healthcare). The mixture of sarcoplasmic proteome and rehydration buffer was loaded onto immobilized pH gradient (IPG) strips (pH 5–8; 17 cm; Bio-Rad). A Protean IEF cell system (Bio-Rad) was used for the first-dimension isoelectric focusing, which enables the separation of proteins based on their isoelectric point. A low voltage (50 V) was applied during the initial active rehydration for 4 h, followed by a rapid increase in voltage, and a final rapid voltage ramping to attain a total of 80 kVh. Subsequently, the IPG strips were equilibrated with equilibration buffer I (6 M urea, 0.375 M Tris-HCl, pH 8.8, 2% sodium dodecyl sulfate [SDS], 20% glycerol, 2% [w/v] dithiothreitol; Bio-Rad) followed by equilibration buffer II (6 M urea, 0.375 M Tris-HCl, pH 8.8, 2% SDS, 20% glycerol, 2.5% [w/v] iodoacetamide; Bio-Rad) for 15 min each. Separation of proteins in the second dimension based on molecular weight was performed using SDS-polyacrylamide gel electrophoresis (SDS-PAGE) in a Protean II Multicell system (Bio-Rad) with 18.5 cm × 20 cm lab cast gels (13.5% acrylamide; 38.5:1 ratio of acrylamide to bis-acrylamide). The equilibrated IPG strips were loaded onto the gels using an agarose overlay, and electrophoresis was completed using running buffer (25 mM Tris, 192 mM glycine, 0.1% SDS) at room temperature. A constant voltage of 100 V was applied for approximately 16 h to allow the separation of proteins in the second dimension. The gel staining was performed using Colloidal Coomassie Blue (20% methanol, 8% ammonium sulfate, 1.9% phosphoric acid, and 0.1% Coomassie Brilliant Blue G-250, Bio-Rad) for 48 h followed by destaining in deionized distilled water for 48 h or until sufficient background clearing was obtained. Each treatment was analyzed in duplicate, resulting in 36 gels.
Gel image analysis
The gel images were obtained using VersaDoc imager (Bio-Rad) and were analyzed using PDQUEST software (version 7.3.1; Bio-Rad). Spots were detected and then matched with the aid of landmarks, which are well-resolved spots present in every gel image. The matched spots were normalized by expressing the relative quantity of each spot (in parts per million) as the ratio of individual spot quantity to the total quantity of valid spots. For each spot in a given sample, spot quantity values in duplicate gels were averaged for statistical analysis. The spots exhibiting a 2.0-fold or more intensity difference between the treatments and associated with 5% statistical significance (P < 0.05) in the Student t test were considered differentially abundant (Joseph et al., 2012; Nair et al., 2018).
Liquid chromatography–electrospray ionization–tandem mass spectrometry
Duplicate spots from the corresponding gels were subjected to mass spectrometric protein identification as described by Nair et al. (2018). The differentially abundant protein spots were excised and subjected to dithiothreitol reduction, iodoacetamide alkylation, and in-gel trypsin digestion. The peptides were extracted, concentrated, and injected for nano-liquid chromatography (LC)–tandem mass spectrometry (MS/MS) analysis using an LTQ Orbitrap mass spectrometer (Thermo Fisher Scientific Inc., Waltham, MA) coupled with an Eksigent Nanoflex cHiPLC system (Eksigent Technologies, Dublin, CA) through a nano-electrospray ionization source. A reverse-phase cHiPLC column (75 μm × 150 mm) operated at a flow rate of 300 nL/min was used for separation of the peptides. The mobile phase A consisted of water with 0.1% (v/v) formic acid, whereas mobile phase B contained acetonitrile with 0.1% (v/v) formic acid. A 50-min gradient was applied: initial 3% mobile phase B was increased linearly to 50% in 24 min and further to 85% and 95% for 5 min each before it was decreased to 3%, and the column was re-equilibrated. The mass analysis method consisted of 8 scan events per segment. The first scan event was an Orbitrap MS scan (100 to 1,600 m/z) with 60,000 resolutions for parent ions, which was followed by data-dependent MS/MS for fragmentation of the 7 most intense ions through collision-induced dissociation.
Protein identification
The LC–MS/MS data were submitted to a local Mascot server for MS/MS protein identification via Proteome Discoverer (version 1.3, Thermo Fisher Scientific, Waltham, MA) against a Bos taurus database from the National Center for Biotechnology Information. Parameters used in the MASCOT MS/MS ion search were as follows: trypsin digest with a maximum of 2 miscleavages, cysteine carbamidomethylation, methionine oxidation, a maximum of 0.001% MS error tolerance, and a maximum of 0.8-Da MS/MS error tolerance. A decoy database was built and searched. Filter settings that determine false discovery rates were used to distribute the confidence indicators for the peptide matches. Peptide matches that passed the filter associated with the strict false discovery rate (target setting of 0.01) were assigned as high confidence. For the MS/MS ion search, proteins with 2 or more high-confidence peptides were considered unambiguous identifications without manual inspection. Proteins identified with one high-confidence peptide were manually inspected and confirmed. The differentially expressed proteins were matched against the STRING database (Szklarczyk et al., 2015) to determine the protein–protein interaction network, wherein the network nodes represented the proteins and the lines indicated functional associations.
Statistical analysis of color data
Nine (n = 9) beef strip loins were utilized in this study, and the experimental unit was a strip loin. The experimental design was a split-plot with strip loin as whole plot and the steaks as the subplots. The instrumental color data were analyzed using the PROC MIXED procedure of SAS version 9.4 (SAS Institute Inc., Cary, NC), and the differences among means were detected using the least significant difference at a 5% level.
Results and Discussion
Instrumental color
An influence of cooking (P < 0.05) was observed for lightness (L* value); raw steaks exhibited lowest (P < 0.05) surface lightness, whereas interiors of steaks cooked to 60°C and 71°C exhibited similar (P > 0.05) lightness (Table 1). The process of cooking promotes structural changes and denaturation of protein, leading to a decrease in water-holding capacity (Aaslyng et al., 2003; Tornberg, 2005; Yu et al., 2017). This in turn, influences the superficial light reflectance, contributing to the increase in lightness of cooked meat compared with raw meat.
| Parameter | Raw | C-60 | C-71 |
|---|---|---|---|
| L* value | 36.77 ± 0.65b | 46.60 ± 0.96a | 47.46 ± 1.08a |
| a* value | 26.52 ± 0.36a | 20.68 ± 0.78b | 10.63 ± 0.65c |
| b* value | 20.94 ± 0.46b | 22.25 ± 0.47a | 18.15 ± 0.14c |
| R630/580 | 6.92 ± 0.25a | 3.80 ± 0.18b | 1.99 ± 0.10c |
| Chroma | 33.81 ± 0.47a | 29.26 ± 0.67b | 21.16 ± 0.35c |
| Hue | 38.27 ± 0.60c | 47.18 ± 1.17b | 59.73 ± 1.53a |
Results are expressed as the mean ± standard error of the mean (SEM). C-60 = longissimus lumborum steaks cooked to 60°C internal endpoint temperature. C-71 = longissimus lumborum steaks cooked to 71°C internal endpoint temperature.
Surface color was measured on raw steaks.
Internal cooked color was measured on C-60 and C-71 steaks.
Means without common superscripts in a row are different (P < 0.05).
R630/580 = reflectance at 630 and 580 nm.
Redness (a* value) of steaks decreased (P < 0.05) as the endpoint temperature increased (Table 1). The C-60 steaks demonstrated greater (P < 0.05) internal redness than the C-71 ones. Denaturation of Mb is primarily responsible for the color change in cooked meats (Bowers et al., 1987; Van Laack et al., 1996; Hunt et al., 1999). Mb denaturation begins between 55°C and 65°C and is almost completed between 75°C and 80°C (Hunt et al., 1999) leading to a decline in redness as the endpoint temperatures increase (Hague et al., 1994; Hunt et al., 1999). In agreement with our results, Suman et al. (2010) observed greater redness for LL steaks cooked to 66°C than those cooked to 71°C. Additionally, a decrease in redness has been documented in beef longissimus thoracis (Yancey et al., 2011), semimembranosus (Yancey et al., 2016), infraspinatus (Yancey et al., 2016), and gluteus medius (John et al., 2005) steaks with an increase in endpoint temperature. Moreover, Rincon et al. (2015) documented a decrease of redness in nonintact beef LL steaks as the cooking temperature increased.
There was an influence (P < 0.05) of endpoint temperature on yellowness (b* values) of the beef steaks (Table 1); steaks cooked to 60° exhibited greatest (P < 0.05) internal yellowness compared with raw steaks and steaks cooked to 71°C. Yancey et al. (2011) reported greater yellowness in longissimus thoracis steaks cooked to 65.5°C than their counterparts cooked to 71.1°C. Moreover, Rincon et al. (2015) documented a decrease of yellowness in nonintact beef LL steaks as the cooking temperature increased from 65°C to 75°C.
R630/580 provides the relative proportion of brown pigments in meat; a lower ratio indicates greater metmyoglobin content and a dull-brown color. Similar to redness, the internal R630/580 values decreased (P < 0.05) with an increase in the endpoint temperature (Table 1). Raw steaks exhibited greater (P < 0.05) R630/580 than the cooked ones; additionally, the interiors of the steaks cooked to 60°C demonstrated greater R630/580 than those cooked to 71°C (Table 1). In agreement with our results, Berry and Bigner-George (2000) reported greater R630/580 values in beef patties cooked to 66°C than their counterparts cooked to 71°C. In addition, Mancini et al. (2010) and Suman et al. (2009) documented a decrease in R630/580 in ground beef patties as the cooking temperature increased from 66°C to 71°C.
Chroma (saturation index; high values are more intense red) demonstrated a trend similar to the redness; the interiors of C-60 steaks demonstrated greater (P < 0.05) chroma than their C-71 counterparts (Table 1). Similarly, Yancey et al. (2011) reported greater chroma in longissimus thoracis steaks cooked to 65.5°C than the steaks cooked to 71.1°C. Furthermore, Hunt et al. (1999) documented greater chroma in beef patties cooked to 65°C than their counterparts cooked to 75°C. Hague et al. (1994) also reported greater chroma in ground beef patties cooked to 60°C than in their counterparts cooked to 71°C.
Conversely, hue angle (trueness of red; high values are more intense brown) was greater in the interiors of steaks cooked to 71°C (P < 0.05) than their counterparts cooked to 60°C (Table 1). As cooking temperature increased, the redness decreased and hue values increased, indicating loss of redness in cooked steaks. In agreement with our results, Yancey et al. (2011) reported greater hue angle in longissimus thoracis steaks cooked to 71.1°C than the steaks cooked to 65.5°C. Moreover, Hunt et al. (1999) documented greater hue values in beef patties cooked to 75°C than their counterparts cooked to 65°C and uncooked patties. In addition, Hague et al. (1994) documented an increase in hue angle in beef patties as endpoint temperature increased from 55°C to 71°C.
Sarcoplasmic proteome analyses
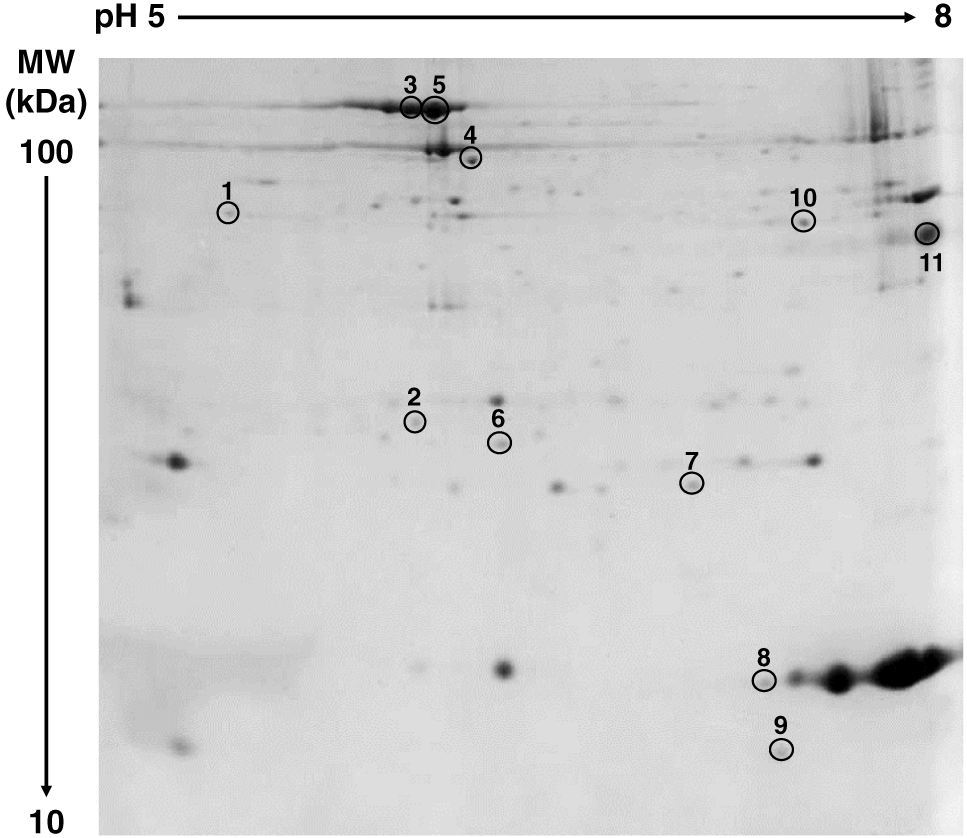
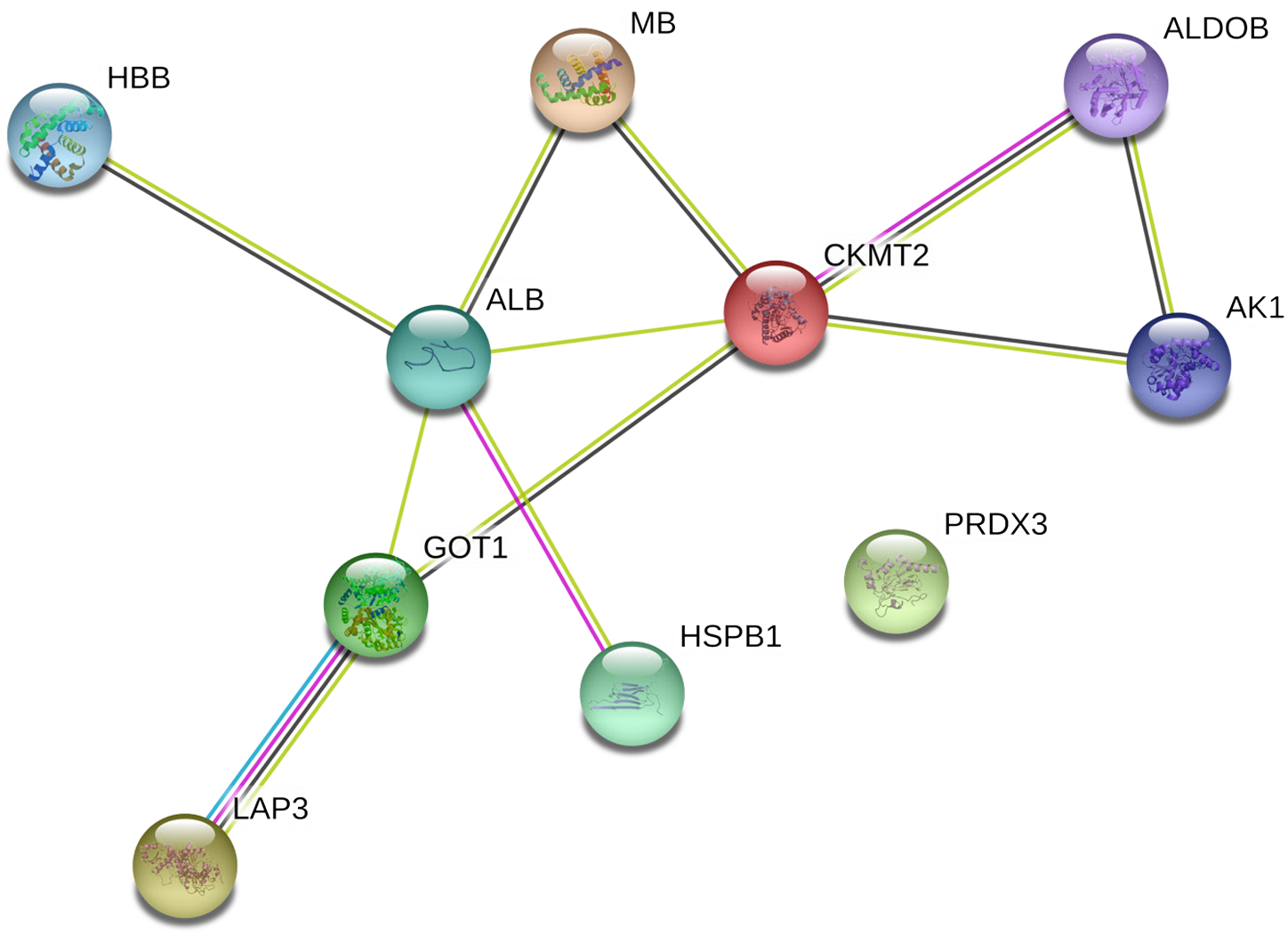
Analyses of gel images indicated that 11 protein spots are differentially abundant (P < 0.05) in the soluble proteome from the C-60 and C-71 steaks. The representative gel image of sarcoplasmic proteome of cooked steak is presented (Figure 1), and the differentially abundant sarcoplasmic protein spots are listed (Table 2). Ten spots were overabundant in C-60 steaks, whereas one spot was overabundant in C-71 samples. In addition, 2 spots (spots 3 and 5; Figure 1) were identified as serum albumin. The differentially abundant proteins could be categorized as enzymes involved in energy metabolism, chaperones, antioxidant proteins, glycolytic enzymes, transport proteins, and enzymes associated with amino acid metabolism. The network of interacting proteins generated using the STRING database (Figure 2) identified 9 proteins as key nodes in biological interactions.
Identity and functional roles of differentially abundant proteins in the sarcoplasmic proteome of longissimus lumborum steaks (n = 9) cooked to 60°C (C-60) and 71°C (C-71) internal endpoint temperatures
| Spota | Protein | Accession number | ProtScore | Sequence coverage (%) | Overabundant treatment | Spot ratiob | Function |
|---|---|---|---|---|---|---|---|
| 1 | Creatine kinase S-type | Q3ZBP1 | 370.42 | 54.65 | C-60 | 1.92 | Energy metabolism |
| 2 | Thioredoxin-dependent peroxide reductase | P35705 | 80.52 | 13.62 | C-60 | 2.56 | Antioxidant |
| 3 | Serum albumin | P02769 | 11079.00 | 80.72 | C-60 | 12.50 | Transport |
| 4 | Cytosol aminopeptidase | P00727 | 6691.23 | 70.13 | C-60 | 4.35 | Amino acid metabolism |
| 5 | Serum albumin | P02769 | 15486.67 | 83.03 | C-60 | 11.1 | Transport |
| 6c | Heat shock protein beta-1 | Q3T149 | 92.10 | 23.38 | C-60 | ND | Chaperone |
| 7 | Adenylate kinase isoenzyme 1 | P00570 | 64.75 | 23.71 | C-60 | 16.67 | Energy metabolism |
| 8 | Myoglobin | P02192 | 2002.63 | 72.73 | C-71 | 0.35 | Oxygen transport |
| 9 | Hemoglobin subunit beta | P02070 | 2438.49 | 88.28 | C-60 | 2.38 | Oxygen transport |
| 10 | Aspartate aminotransferase | P33097 | 2943.29 | 79.66 | C-60 | 2.04 | Amino acid metabolism |
| 11 | Fructose-bisphosphate aldolase B | Q3T0S5 | 38.11 | 1.92 | C-60 | 2.78 | Glycolytic enzyme |
C-60 = longissimus lumborum steaks cooked to 60°C internal endpoint temperature. C-71 = longissimus lumborum steaks cooked to 71°C internal endpoint temperature.
Spot number refers to the numbered spots in gel image (Figure 1). Spots are identified by accession number (UniProt), ProtScore, and sequence coverage of peptides.
Spot ratio of C-60/C-71.
Protein absent in C-71.
ND = not detected.
Protein–protein interaction network of differentially abundant proteins in the sarcoplasmic proteome of longissimus lumborum steaks cooked to 60°C and 71°C internal endpoint temperature. The interacting proteins were identified using STRING 11.0 software (Szklarczyk et al., 2015). The nodes represent proteins from a Bos taurus database, whereas the lines (purple = experimental evidence; light green = text mining evidence; black = co-expression evidence; light blue = evidence from curated databases) indicate predicted functional annotations. ALB = serum albumin; ALDOB = fructose-bisphosphate aldolase B; AK1 = adenylate kinase isoenzyme 1; CKMT2 = creatine kinase S-type; GOT1 = aspartate aminotransferase; HBB = hemoglobin subunit beta; HSPB1 = heat shock protein beta-1; LAP3 = cytosol aminopeptidase; MB = myoglobin; PRDX3 = thioredoxin-dependent peroxide reductase.
The greater abundance of 10 protein spots in steaks cooked to 60°C could be attributed to the lower endpoint temperature during cooking (Tornberg, 2005; Sarah et al., 2014; Yu et al., 2016). Heating induces several structural changes in sarcoplasmic proteins, which aggregate at temperatures between 40°C and 60°C (Tornberg, 2005; Wen et al., 2015; Yu et al., 2017) and up to 90°C (Tornberg, 2005). As the temperature increases, the proteins start to unfold, losing their tertiary and secondary structures (Yu et al., 2017). This in turn promotes denaturation and aggregation of proteins, reducing their extractability (Dai et al., 2013; Yu et al., 2016) as well as staining intensity in the gel (Montowska and Pospiech, 2013; Sarah et al., 2014; Yu et al., 2017). Wen et al. (2015) reported that SDS-PAGE gel band intensities of proteins extracted decreased as the internal cooking temperature of pork increased from 60°C to 100°C, which was attributed to the protein aggregation.
Previous proteomic investigations have also reported differential abundance and heat tolerance of sarcoplasmic proteins in cooked meat. Montowska and Pospiech (2013) compared the sarcoplasmic proteome profile of raw meat and cooked (at 75°C) meat from different species (cattle, pig, chicken, turkey, duck, and goose) and reported the thermal resistance of transport proteins and chaperones. Similarly, Li et al. (2018) reported the thermal stability of Mb, hemoglobin, serum albumin, and creatine kinase in meat from 5 species (beef, sheep, pig, duck, and chicken) cooked using different methods/temperatures (water bath at 78°C, boiling at 100°C, sterilizing at 121°C, frying and baking at 200°C). Tian et al. (2016) performed proteomic analysis of beef LL muscle cooked with 3 different methods (water bath, short-time ohmic cooking, and long-time ohmic cooking) at 72°C and reported thermal stability of heat shock proteins and serum albumin.
Enzymes involved in energy metabolism
Creatine kinase S-type and adenylate kinase isoenzyme 1 are the enzymes involved in energy metabolism and were more abundant (P < 0.05) in C-60 steaks compared with C-71 steaks (Table 2). Previous research (Bai et al., 1998; He et al., 2005; Fedurkina et al., 2006) reported that creatine kinase begins to denature at temperatures near 50°C and is completely unfolded around 60°C. This, in turn, could explain the overabundance of this protein in C-60 steaks compared with C-71 counterparts. Creatine kinase S-type is a mitochondrial creatine kinase isoenzyme, which reversibly catalyzes the interconversion of adenosine diphosphate (ADP) and phosphocreatine to adenosine triphosphate (ATP) and creatine, contributing to the ATP–ADP equilibrium in postmortem skeletal muscles (Gao et al., 2016; Zhai et al., 2018). Creatine kinase S-type has rarely been previously reported in meat research. However, the overabundance of creatine kinase M-type in beef steaks has been related to improved color stability (Joseph et al., 2012; Canto et al., 2015; Nair et al., 2018). Creatine kinase M-type can increase the creatine content, which acts as an antioxidant to minimize Mb oxidation and contribute to improve color stability (Canto et al., 2015). Heat-induced aggregation of creatine kinase M-type at temperatures above 60°C has been documented in rabbit (Fedurkina et al., 2006) and lamb (Yu et al., 2015) muscles.
Adenylate kinase isoenzyme 1 catalyzes the reversible conversion of 2 molecules of ADP to ATP and adenosine monophosphate (Heil et al., 1974; Janssen et al., 2003) and was overabundant (P <0.05) in the steaks cooked to 60°C compared with in those cooked to 71°C. Zhang et al. (1993) documented that the thermal inactivation of adenylate kinase isoenzyme 1 occurs between 35°C and 60°C, which in turn could possibly explain the overabundance of this protein in C-60 steaks. The abundance of this protein has been previously associated with the color stability of fresh beef (Canto et al., 2015; Wu et al., 2015a). However, the exact mechanism through which this enzyme influences meat color is not clearly understood. Wu et al. (2015a) documented a positive correlation of adenylate kinase isoenzyme 1 and metmyoglobin reducing activity in LL muscle from Chinese yellow cattle. In contrast, Canto et al. (2015) attempted to explain animal-to-animal variation in beef color stability and documented the overabundance of adenylate kinase isoenzyme 1 in color-labile LL muscles compared with in color-stable LL muscles. Hwang et al. (2005) observed that 3 proteins (adenylate kinase, troponin T, and ATP-dependent proteinase SP-22) were negatively correlated to drip loss in porcine longissimus muscle and suggested that the degradation of these proteins increased drip loss, consequently affecting reflectance and resulting in lighter appearance of meat.
Chaperones
Heat shock protein beta-1 (HSP27) belongs to the family of small heat shock proteins that plays a critical role in the stabilization of the cytoskeleton of stressed cells (Perng et al., 1999; Bryantsev et al., 2007). In addition, the small heat shock proteins can act as ATP-independent molecular chaperones, binding to partially unfolded proteins and preventing their aggregation (Bryantsev et al., 2007). The HSP27 was present only in steaks cooked to 60°C (Table 2). The absence of this protein in steaks cooked to 71°C could be attributed to the increase in the internal endpoint temperature. Pivovarova et al. (2005) documented that the denaturation of HSP27 occurs at temperatures around 70°C, and cooking temperatures above it (i.e., 71°C) could influence the conformational changes of the protein, leading to unfolding and aggregation (Tornberg, 2005) decreasing their extractability and staining intensity in the gels (Dai et al., 2013; Montowska and Pospiech, 2013; Tian et al., 2016). The abundance of HSP27 have been previously associated with fresh meat color (Joseph et al., 2012; Wu et al., 2015a). Joseph et al. (2012) evaluated the differential abundance of sarcoplasmic proteome in color-labile and color-stable beef muscles and documented a positive correlation between HSP27 and color stability. Conversely, Wu et al. (2015a) documented a negative correlation of HSP27 with redness in LL muscle from Chinese yellow cattle. In cooked meat, the heat shock proteins provide protection to proteins against structural changes by preventing denaturation and aggregation (Pivovarova et al., 2005), which in turn could contribute to Mb stability upon cooking (Tian et al., 2016).
Antioxidant proteins
Thioredoxin-dependent peroxide reductase belongs to a group of antioxidant enzymes known as peroxiredoxins. Peroxiredoxins are cysteine-dependent peroxidase enzymes, which play a critical role in reducing peroxides in cells (Wood et al., 2003; Joseph et al., 2012). Mammalian peroxiredoxins have 6 isomers. Thioredoxin-dependent peroxide reductase is a 2-cysteine peroxiredoxin abundant in mitochondria, and its main function is to reduce the natural hydroperoxides through peroxidase activity (Wood et al., 2003). Thioredoxin-dependent peroxide reductase was more abundant (P < 0.05) in steaks cooked to 60°C compared with those cooked to 71°C, and this observation could be attributed to the differences in endpoint temperature. Liau et al. (2010) examined the thermal stability of peroxiredoxins and reported that these antioxidant proteins lose 40% of their activity after heating at 60°C for 2.5 min. In our study, steaks were cooked approximately for 10 min prior to reaching desired internal temperatures, which in turn could have accelerated the denaturation of thioredoxin-dependent peroxide reductase leading to their overabundance in C-60 steaks compared with C-71 steaks. Previous investigations have documented the relationship between peroxiredoxins and fresh meat color. Joseph et al. (2012) evaluated the differential abundance of sarcoplasmic proteome in color-stable (LL) and color-labile (psoas major) beef muscles and observed a positive correlation of peroxiredoxin with color stability. In addition, Wu et al. (2015a) documented a positive correlation between peroxiredoxin with surface redness and metmyoglobin reducing activity in LL muscle from Chinese yellow cattle. Similarly, Gagaoua et al. (2015) documented that peroxiredoxin was positively correlated to redness in longissimus thoracis muscle from Blond d’Aquitaine bulls, which may be explained by the antioxidant and phospholipase activities of this protein. The role of peroxiredoxin in eliminating peroxides could contribute to the minimization of lipid oxidation. This, in turn, minimizes lipid oxidation–induced Mb oxidation (Faustman et al., 2010) and thus improves beef color stability (Suman and Joseph, 2013; Suman et al., 2014). Furthermore, Promeyrat et al. (2011) reported a negative correlation between the intensity of peroxiredoxins and protein oxidation in porcine LL cooked to 100°C, demonstrating a potential protection offered by peroxiredoxins against heat-induced protein damages.
Glycolytic enzyme
Fructose-bisphosphate aldolase B, an enzyme that catalyzes the reversible cleavage of fructose 1,6-bisphosphate to dihydroxyacetone phosphate and glyceraldehyde 3-phosphate (Marsh and Lebherz, 1992; Esposito et al., 2002), is found in 3 different isoforms—muscle type (aldolase A), liver type (aldolase B), and brain type (aldolase C)—in vertebrates (Esposito et al., 2002). This enzyme was more abundant (P < 0.05) in C-60 steaks compared with their C-71 counterparts (Table 2). This could be partially explained by the protein’s thermal stability; the melting temperature of aldolase B has been reported to be below 50°C (Esposito et al., 2002). Several studies have reported the association between fructose-bisphosphate aldolase and fresh meat color (Wu et al., 2015a, 2015b; Nair et al., 2016). Wu et al. (2015a, 2015b) documented that the abundance of fructose-bisphosphate aldolase A exhibited a negative correlation with redness in LL from Chinese Luxi cattle. Similarly, Nair et al. (2016) attributed the greater abundance of glycolytic enzymes (i.e., fructose-bisphosphate aldolase A) to the rapid postmortem glycolysis and to the lower color stability of color-labile inside beef semimembranosus muscle. In addition, previous studies reported that fructose-bisphosphate aldolase A was involved in cooking-induced protein aggregation in lamb longissimus thoracis et lumborum boiled at 100°C (Yu et al., 2015) and roasted at 190°C (Yu et al., 2016).
Transport proteins
The differentially abundant transport proteins included hemoglobin subunit beta, Mb, and serum albumin. Hemoglobin subunit beta was overabundant (P < 0.05) in the C-60 steaks compared with C-71 steaks (Table 2). Yan et al. (2004) reported that hemoglobin undergoes thermal unfolding at 44°C–54°C and is completely aggregated at 54°C–70°C, which in turn could explain the overabundance of this heme protein in steaks cooked to 60°C compared with those cooked to 71°C. Previous studies reported that hemoglobin can be utilized as a biomarker in cooked meats for species authentication (Montowska, 2017; Fornal and Montowska, 2019). Although bleeding removes the majority of blood from the food animals, some residual blood could remain inside the arteries and veins within the skeletal muscles, resulting in the presence of hemoglobin in meat (Suman and Joseph, 2013). Nonetheless, the contribution of hemoglobin to meat color is only minimal (Faustman and Cassens, 1990; Suman and Joseph, 2013).
Mb was identified as the protein in a differentially abundant spot (spot 8 in Figure 1), and it was the only protein spot more abundant (P < 0.05) in the steaks cooked to 71°C than in the ones cooked to 60°C (Table 2). While this interesting observation was unexpected and is not readily explained, it could be probably due to the increased thermal stability of Mb as a result of post-translational modifications. Our previous study documented that beef Mb appears in two-dimensional electrophoresis gels at multiple spots, characterized by different isoelectric pH but similar molecular weight (Canto et al., 2015), which has been attributed to post-translational modifications. Additionally, Canto et al. (2015) observed that one Mb spot was more abundant in color-labile beef LL raw steaks than in the color-stable LL steaks, despite similar Mb concentration in color-labile and color-stable steaks. Post-translational modifications such as glycosylation (Locke et al., 2006), methylation (Kumar et al., 2004), acetylation (Zhu et al., 2005; Lengqvist et al., 2011), and phosphorylation (Maurides et al., 1989; Zhu et al., 2005; Anderson et al., 2014) can lead to changes in isoelectric pH of proteins with minimal impact on molecular mass. In this perspective, the differentially abundant Mb spot in cooked steaks observed in the present study could possibly be the result of post-translational modifications since there are several other spots closer to it at same molecular weight but with more alkaline isoelectric pH (Figure 1).
Post-translational modifications have been reported to affect thermal stability of fish (Huang and Ochiai, 2005) and mouse (Chetsanga and Liskiwskyi, 1977) muscle proteins. Additionally, Alderton et al. (2003) documented that the alkylation influenced thermal stability of beef Mb. Recently, Mitra et al. (2018) observed that several proteins, including Mb, in pork longissimus muscle underwent post-translational modifications upon cooking. The observed overabundance of an Mb spot in the steaks cooked to 71°C may be due to the fact that post-translational modifications (induced by heating above 60°C) could have possibly led to an increase in thermal stability. Nonetheless, we did not confirm post-translational modifications in Mb to verify this possibility. Further research is necessary to examine the role of Mb post-translational modifications in meat color.
Although Mb undergoes denaturation at various degrees in cooked meats, it has been reported as a heat-stable protein in meats (Janssen et al., 1990; Taylor et al., 1993; Ponce et al., 1994; Yu et al., 2017). Sarah et al. (2014) examined the effect of different heat treatments (boiled at 100°C and autoclaved at 121°C) on proteins in goat longissimus thoracis and reported Mb among the proteins that exhibited high heat stability. Furthermore, the presence of heat-stable Mb peptides has been documented in cooked lamb (Watson et al., 2015; Yu et al., 2016) and beef (Montowska et al., 2014, 2015; Watson et al., 2015; Li et al., 2018).
Serum albumin was identified in 2 spots (spots 3 and 5 in Figure 1), and both these spots were overabundant (P < 0.05) in the C-60 steaks compared with C-71 ones (Table 2). The two spots identified as serum albumin demonstrated different isoelectric pH but similar molecular weight (Figure 1). The variations in the isoelectric point of serum albumin could be attributed to post-translational modifications, which lead to shifts in the isoelectric pH (Gianazza, 1995; Halligan et al., 2004; Lengqvist et al., 2011). Serum albumin is a transport and antioxidant protein, playing a main role in regulation of colloidal osmotic pressure of blood (Roche et al., 2008). The overabundance of serum albumin in the C-60 steaks could be partially attributed to its denaturation temperature, which falls in the range of 50°C (Lu et al., 2015) to 71ºC (Gulseren et al., 2007). The heat-induced denaturation could accelerate the insolubility and aggregation of proteins, resulting in a lower sarcoplasmic protein extractability (Dai et al., 2013; Montowska and Pospiech, 2013; Tian et al., 2016). The presence of serum albumin may be due to low levels blood in muscle after bleeding (Montowska and Pospiech, 2013). Furthermore, serum albumin was identified in sarcoplasmic proteins extracted from the muscles of cattle (Bouley et al., 2004). Several previous proteomic investigations documented the presence of serum albumin in cooked meats. In their attempts to identify heat-stable biomarkers for meats, Li et al. (2018) reported the presence of serum albumin peptide in cooked lamb. Montowska and Pospiech (2013) compared the sarcoplasmic proteome profile of meat from different species (cattle, pig, chicken, turkey, duck, and goose) and identified serum albumin in beef cooked to 75°C. Moreover, Sarah et al. (2016) documented a peptide from serum albumin in pork cooked with 2 different methods (boiling at 100°C and autoclaving at 121°C).
Enzymes involved in amino acid metabolism
Cytosol aminopeptidase, also known as leucine aminopeptidase, is an enzyme that catalyzes the removal of N-terminal amino acids from the peptides and is involved in the regular turnover of intracellular proteins (Wu et al., 2015b) at the final step in protein degradation (Flores et al., 1996; Matsui et al., 2006). Aminopeptidases found in cytosol of skeletal muscle (Toldra et al., 1992; Flores et al., 1996, 1998) play a major role in the degradation of peptides, generated by endopeptidases, into free amino acids (Nishimura et al., 1991; Flores et al., 1996). This enzyme was more abundant (P < 0.05) in steaks cooked to 60°C compared with their counterparts cooked to 71°C (Table 2). Matsushita-Morita et al. (2011) reported that the activity of cytosol aminopeptidase remained stable at temperatures up to 60°C, and the enzyme became completely inactive at 70°C. The differences in endpoint temperature (60°C vs. 71°C) could have led to the denaturation/inactivation of this enzyme and contributed to its differential abundance. Cytosol aminopeptidase has been reported previously in chicken (Nishimura et al., 1991), pork (Bax et al., 2013) and beef (Bouley et al., 2004) skeletal muscles. Proteomic studies documented the involvement of this protein in tenderization of beef longissimus muscle during aging (Polati et al., 2012). Furthermore, cytosol aminopeptidase exhibited a positive correlation with redness and metmyoglobin reducing activity and a negative correlation with lipid oxidation in semitendinosus muscle of Chinese yellow cattle (Wu et al., 2015b). However, the exact mechanism through which this enzyme influences the color of fresh and cooked meat is not clearly understood.
Aspartate aminotransferase, also known as glutamic-oxaloacetic transaminase 1, is a cytoplasmic enzyme catalyzing the reversible transfer of an amino group between aspartate and glutamate (Aulbach and Amuzie, 2017) and thus plays a major role in amino acid metabolism (Gallego et al., 2016; Sayd et al., 2016). This enzyme was more abundant (P < 0.05) in C-60 steaks than in C-71 ones, and this observation could be partially explained based on its thermal stability. Hamm et a1. (1969) documented that the optimum temperature for activity of aspartate aminotransferase in bovine muscle was around 50°C, and the rate of its maximum decline occurred between 71.1°C and 75.6°C (Searcy et al., 1995). The increase of endpoint temperature can lead to the unfolding and inactivation of aspartate aminotransferase (Adams, 1991), which in turn could have contributed to its overabundance in C-60 steaks. Proteomic studies have reported the presence of this enzyme in broiler breast muscle (Kuttappan et al., 2017) and in dry-cured ham (Gallego et al., 2016). Nonetheless, the role of this protein in meat quality remains unknown.
Conclusions
The internal cooked color and sarcoplasmic proteome of beef LL steaks were influenced by endpoint temperature. Steaks cooked to 60°C demonstrated greater redness and color stability than their counterparts cooked to 71°C. Antioxidant proteins, glycolytic enzymes, transport proteins, proteins associated with energy metabolism, and chaperones were differentially abundant between the C-60 and C-71 steaks. The differential abundance of sarcoplasmic proteome components indicated that the overabundant proteins in the C-60 steaks may be utilized as potential biomarkers for undercooked beef. Additionally, possible post-translational modification of Mb in cooked steaks appeared to influence the heme protein’s thermal stability. Further work should examine the role of post-translational modifications of Mb in cooked meat color so that biomarkers can be identified for quality defects in cooked meats.
Acknowledgements
This work was supported by the National Council for Scientific and Technological Development (CNPq), Brazil (Grant Numbers 400136/2014-7 and 311422/2016-0); the Carlos Chagas Filho Research Foundation (FAPERJ), Brazil (Grant Numbers E-26/010.001703/2015, E-26/010.001547/2016, and E-26/203.049/2017); and the Coordination for the Improvement of Higher Education Personnel (CAPES), Brazil (Financing Code 001). A. P. A. A. Salim was supported by a CNPq doctorate fellowship (141838/2015-9). The authors also thank CAPES, Brazil, for providing the PDSE scholarship (88881.133504/2016-01) to A. P. A. A. Salim for completing doctoral research at the University of Kentucky. S. P. Suman was supported by a CNPq Special Visiting Researcher Fellowship (303477/2014-8) through the Science without Borders program. This work was also supported by the National Institute of Food and Agriculture, U.S. Department of Agriculture, Hatch-Multistate Project 1014747.
Literature Cited
Aaslyng, M. D., C. Bejerholm, P. Ertbjerg, H. C. Bertram, and H. J. Andersen. 2003. Cooking loss and juiciness of pork in relation to raw meat quality and cooking procedure. Food Qual. Prefer. 14:277–288. https://doi.org/10.1016/S0950-3293(02)00086-1.
Adams, J. B. 1991. Review: Enzyme inactivation during heat processing of food-stuffs. Int. J. Food Sci. Tech. 26:1–20. https://doi.org/10.1111/j.1365-2621.1991.tb01136.x.
Alderton, A. L., C. Faustman, D. C. Liebler, and D. W. Hill. 2003. Induction of redox instability of bovine myoglobin by adduction with 4-hydroxy-2-nonenal. Biochemistry-US. 42:4398–4405. https://doi.org/10.1021/bi0271695.
AMSA. 2012. Meat color measurement guidelines. 2nd edition. American Meat Science Association, Champaign, IL.
Anderson, M. J., S. M. Lonergan, and E. Huff-Lonergan. 2014. Differences in phosphorylation of phosphoglucomutase 1 in beef steaks from the longissimus dorsi with high or low star probe values. Meat Sci. 96:379–384. https://doi.org/10.1016/j.meatsci.2013.07.017.
Aulbach, A. D., and C. J. Amuzie. 2017. Biomarkers in nonclinical drug development. In: A. S. Faqi, editor, A comprehensive guide to toxicology in nonclinical drug development. Academic Press. p. 447–471. https://doi.org/10.1016/B978-0-12-803620-4.00017-7.
Bai, J. H., S. Y. Zheng, and H. M. Zhou. 1998. Inactivation of creatine kinase is due to the conformational changes of the active sites during thermal denaturation. Biochem. Mol. Biol. Int. 45:941–951. https://doi.org/10.1002/iub.7510450512.
Bax, M. L., T. Sayd, L. Aubry, C. Ferreira, D. Viala, C. Chambon, D. Remond, and V. Sante-Lhoutellier. 2013 Muscle composition slightly affects in vitro digestion of aged and cooked meat: Identification of associated proteomic markers. Food Chem. 136:1249–1262. https://doi.org/10.1016/j.foodchem.2012.09.049.
Berry, B. W, and M. E. Bigner-George. 2000. Factors affecting color properties of beef patties cooked on an outdoor gas grill. J. Muscle Foods. 11:213–226. https://doi.org/10.1111/j.1745-4573.2000.tb00426.x.
Bouley, J., C. Chambon, and B. Picard. 2004. Mapping of bovine skeletal muscle proteins using two-dimensional gel electrophoresis and mass spectrometry. Proteomics. 4:1811–1824. https://doi.org/10.1002/pmic.200300688.
Bowers, J. A., J. A. Craig, D. H. Kropf, and T. J. Tucker. 1987. Flavor, color, and other characteristics of beef longissimus muscle heated to seven internal temperatures between 55° and 85°C. J. Food Sci. 52:533–536. https://doi.org/10.1111/j.1365-2621.1987.tb06668.x.
Bradford, M. M. 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3.
Bryantsev, A. L., S. Y. Kurchashova, S. A. Golyshev, V. Y. Polyakov, H. F. Wunderink, B. Kanon, K. R. Budagova, A. E. Kabakov and H. H. Kampinga. 2007. Regulation of stress-induced intracellular sorting and chaperone function of Hsp27 (HspB1) in mammalian cells. Biochem. J. 407:407–417. https://doi.org/10.1042/BJ20070195.
Canto, A. C. V. C. S., S. P. Suman, M. N. Nair, S. Li, G. Rentfrow, C. M. Beach, T. J. P. Silva, T. L. Wheeler, S. D. Shackelford, A. Grayson, R. O. McKeith, and D. A. King. 2015. Differential abundance of sarcoplasmic proteome explains animal effect on beef Longissimus lumborum color stability. Meat Sci. 102:90–98. https://doi.org/10.1016/j.meatsci.2014.11.011.
Chetsanga, C. J., and M. Liskiwskyi. 1977. Decrease in specific activity of heart and muscle aldolase in old mice. Int. J. Biochem. 8:753–765. https://doi.org/10.1016/0020-711X(77)90027-1.
Dai, Y., J. Miao, S. Z. Yuan, Y. Liu, X. M. Li, and R. T. Dai. 2013. Colour and sarcoplasmic protein evaluation of pork following water bath and ohmic cooking. Meat Sci. 93:898–905. https://doi.org/10.1016/j.meatsci.2012.11.044.
Esposito, G., L. Vitagliano, R. Santamaria, A. Viola, A. Zagari, and F. Salvatore. 2002. Structural and functional analysis of aldolase B mutants related to hereditary fructose intolerance. FEBS Lett. 531:152–156. https://doi.org/10.1016/S0014-5793(02)03451-8.
Faustman, C., and R. G. Cassens. 1990. The biochemical basis for discoloration in fresh meat: A review. J. Muscle Foods. 1:217–243. https://doi.org/10.1111/j.1745-4573.1990.tb00366.x.
Faustman, C., Q. Sun, R. Mancini, and S. P. Suman. 2010. Myoglobin and lipid oxidation interactions: Mechanistic bases and control. Meat Sci. 86:86–94. https://doi.org/10.1016/j.meatsci.2010.04.025.
Fedurkina, N. V., L. V. Belousova, L. G. Mitskevich, H. M. Zhou, Z. Chang, and B. I. Kurganov. 2006. Change in kinetic regime of protein aggregation with temperature increase. Thermal aggregation of rabbit muscle creatine kinase. Biochemistry-Moscow+. 71:325–331. https://doi.org/10.1134/S000629790603014X.
Flores, M., M. C. Aristoy, and F. Toldra. 1996. HPLC Purification and characterization of soluble alanyl aminopeptidase from porcine skeletal muscle. J. Agr. Food Chem. 44:2578–2583. https://doi.org/10.1021/jf9504677.
Flores, M., M. C. Aristoy, and F. Toldra. 1998. Feedback inhibition of porcine muscle alanyl and arginyl aminopeptidases in cured meat products. J. Agr. Food Chem. 46:4982–4986. https://doi.org/10.1021/jf9806863.
Fornal, E., and M. Montowska. 2019. Species-specific peptide-based liquid chromatography–mass spectrometry monitoring of three poultry species in processed meat products. Food Chem. 283:489–498. https://doi.org/10.1016/j.foodchem.2019.01.074.
Gagaoua, M., B. Picard, and V. Monteils. 2018. Associations among animal, carcass, muscle characteristics, and fresh meat color traits in Charolais cattle. Meat Sci. 140:145–156. https://doi.org/10.1016/j.meatsci.2018.03.004.
Gagaoua, M., E. M. C. Terlouw, D. Micol, A. Boudjellal, J. F. Hocquette, and B. Picard. 2015. Understanding early post-mortem biochemical processes underlying meat color and pH decline in the longissimus thoracis muscle of young Blond d’Aquitaine bulls using protein biomarkers. J. Agr. Food Chem. 63:6799–6809. https://doi.org/10.1021/acs.jafc.5b02615.
Gagaoua, M., E. C. Terlouw, and B. Picard. 2017. The study of protein biomarkers to understand the biochemical processes underlying beef color development in young bulls. Meat Sci. 134:18–27. https://doi.org/10.1016/j.meatsci.2017.07.014.
Gallego, M., L. Mora, M. C. Aristoy, and F. Toldra. 2016. The use of label-free mass spectrometry for relative quantification of sarcoplasmic proteins during the processing of dry-cured ham. Food Chem. 196:437–444. https://doi.org/10.1016/j.foodchem.2015.09.062.
Gao, X., W. Wu, C. Ma, X. Li, and R. Dai. 2016. Postmortem changes in sarcoplasmic proteins associated with color stability in lamb muscle analyzed by proteomics. Eur. Food Res. Technol. 242:527–535. https://doi.org/10.1007/s00217-015-2563-2.
Gianazza, E. 1995. Isoelectric focusing as a tool for the investigation of post-translational processing and chemical modifications of proteins. J. Chromatogr. A. 705:67–87. https://doi.org/10.1016/0021-9673(94)01251-9.
Gulseren, I., D. Guzey, B. D. Bruce, and J. Weiss. 2007. Structural and functional changes in ultrasonicated bovine serum albumin solutions. Ultrason. Sonochem. 14:173–183. https://doi.org/10.1016/j.ultsonch.2005.07.006.
Hague, M. A., K. E. Warren, M. C. Hunt, D. H. Kropf, C. L. Kastner, S. L. Stroda, and D. E. Johnson. 1994. Endpoint temperature, internal cooked color, and expressible juice color relationships in ground beef patties. J. Food Sci. 59:465–470. https://doi.org/10.1111/j.1365-2621.1994.tb05539.x.
Halligan, B. D., V. Ruotti, W. Jin, S. Laffoon, S. N. Twigger, and E. A. Dratz. 2004. ProMoST (Protein Modification Screening Tool): A web-based tool for mapping protein modifications on two-dimensional gels. Nucleic Acids Res. 32:W638–W644. https://doi.org/10.1093/nar/gkh356.
Hamm, R., L. Kormendy, and G. Gantner. 1969. Transaminases of skeletal muscle. 1. the activity of transaminasines in post-mortem bovine and porcine muscles. J. Food Sci. 34:446–448. https://doi.org/10.1111/j.1365-2621.1969.tb12801.x.
He, H. W., J. Zhang, H. M. Zhou, and Y. B. Yan. 2005. Conformational change in the c-terminal domain is responsible for the initiation of creatine kinase thermal aggregation. Biophys. J. 89:2650–2658. https://doi.org/10.1529/biophysj.105.066142.
Heil, A., G. Muller, L. Noda, T. Pinder, H. Schirmer, I. Schirmer, and I. Von Zabern. 1974. The amino-acid sequence of porcine adenylate kinase from skeletal muscle. FEBS J. 43:131–144. https://doi.org/10.1111/j.1432-1033.1974.tb03393.x.
Huang, M. C., and Y. Ochiai. 2005. Fish fast skeletal muscle tropomyosins show species-specific thermal stability. Comp. Biochem. Phys. B. 141:461–471. https://doi.org/10.1016/j.cbpc.2005.05.008.
Hunt, M. C., O. Sorheim, and E. Slinde. 1999. Color and heat denaturation of myoglobin forms in ground beef. J. Food Sci. 64:847–51. https://doi.org/10.1111/j.1365-2621.1999.tb15925.x.
Hwang, I. H., B. Y. Park, J. H. Kim, S. H. Cho, and J. M. Lee. 2005. Assessment of postmortem proteolysis by gel-based proteome analysis and its relationship to meat quality traits in pig longissimus. Meat Sci. 69:79–91. https://doi.org/10.1016/j.meatsci.2004.06.019.
Janssen, E., A. D. De Groof, M. Wijers, J. Fransen, P. P. Dzeja, A. Terzic, and B. Wieringa. 2003. Adenylate kinase 1 deficiency induces molecular and structural adaptations to support muscle energy metabolism. J. Biol. Chem. 278:12937–12945. https://doi.org/10.1074/jbc.M211465200.
Janssen, F. W., G. H. Hagele, A. M. B. Voorpostel, and J. A. De Baaij. 1990. Myoglobin analysis for determination of beef, pork, horse, sheep, and kangaroo meat in blended cooked products. J. Food Sci. 55:1528–1530. https://doi.org/10.1111/j.1365-2621.1990.tb03560.x.
John, L., D. Cornforth, C. E. Carpenter, O. Sorheim, B. C. Pettee, and D. R. Whittier. 2005. Color and thiobarbituric acid values of cooked top sirloin steaks packaged in modified atmospheres of 80% oxygen, or 0.4% carbon monoxide, or vacuum. Meat Sci. 69:441–449. https://doi.org/10.1016/j.meatsci.2004.08.013.
Joseph, P., S. P. Suman, G. Rentfrow, S. Li, and C. M. Beach. 2012. Proteomics of muscle-specific beef color stability. J. Agr. Food Chem. 60:3196–3203. https://doi.org/10.1021/jf204188v.
King, N. J., and R. Whyte. 2006. Does it look cooked? A review of factors that influence cooked meat color. J. Food Sci. 71:R31–R40. https://doi.org/10.1111/j.1750-3841.2006.00029.x.
Kumar, Y., A. Khachane, M. Belwal, S. Das, K. Somsundaram, and U. Tatu. 2004. ProteoMod: A new tool to quantitate protein post-translational modifications. Proteomics. 4:1672–1683. https://doi.org/10.1002/pmic.200300778.
Kuttappan, V. A., W. Bottje, R. Ramnathan, S. D. Hartson, C. N. Coon, B. W. Kong, C. M. Owens, M. Vazquez-Anon, and B. M. Hargis. 2017. Proteomic analysis reveals changes in carbohydrate and protein metabolism associated with broiler breast myopathy. Poultry Sci. 96:2992–2999. https://doi.org/10.3382/ps/pex069.
Lengqvist, J., H. Eriksson, M. Gry, K. Uhlen, C. Bjorklund, B. Bjellqvist, P. J. Jakobsson, and J. Lehtio. 2011. Observed peptide pI and retention time shifts as a result of post-translational modifications in multidimensional separations using narrow-range IPG-IEF. Amino Acids. 40:697–711. https://doi.org/10.1007/s00726-010-0704-2.
Li, Y., Y. Zhang, H. Li, W. Zhao, W. Guo, and S. Wang. 2018. Simultaneous determination of heat stable peptides for eight animal and plant species in meat products using UPLC-MS/MS method. Food Chem. 245:125–131. https://doi.org/10.1016/j.foodchem.2017.09.066.
Liau, Y. J., Y. T. Chen, C. Y. Lin, J. K. Huang, and C. T. Lin. 2010. Characterisation of 2-Cys peroxiredoxin isozyme (Prx1) from Taiwanofungus camphorata (Niu-chang-chih): Expression and enzyme properties. Food Chem. 119:154–160. https://doi.org/10.1016/j.foodchem.2009.06.008.
Locke, D., I. V. Koreen, A. L. Harris, D. Locke, I. V. Koreen, and A. L. Harris. 2006. Isoelectric points and post-translational modifications of connexin26 and connexin32. FASEB J. 20:1221–1223. https://doi.org/10.1096/fj.05-5309fje.
Lu, R., W. W. Li, A. Katzir, Y. Raichlin, H. Q. Yu, and B. Mizaikoff. 2015. Probing the secondary structure of bovine serum albumin during heat-induced denaturation using mid-infrared fiberoptic sensors. Analyst. 140:765–770. https://doi.org/10.1039/c4an01495b.
Mahmood, S., N. Turchinsky, F. Paradis, W. T. Dixon, and H. L. Bruce. 2018. Proteomics of dark cutting longissimus thoracis muscle from heifer and steer carcasses. Meat Sci. 137:47–57. https://doi.org/10.1016/j.meatsci.2017.11.014.
Mancini, R. A., and M. C. Hunt. 2005. Current research in meat color. Meat Sci. 71:100–121. https://doi.org/10.1016/j.meatsci.2005.03.003.
Mancini, R. A., R. Ramanathan, S. P. Suman, M. K. R. Konda, P. Joseph, G. A. Dady, B. M. Naveena, and I. Lopez-Lopez. 2010. Effects of lactate and modified atmospheric packaging on premature browning in cooked ground beef patties. Meat Sci. 85:339–346. https://doi.org/10.1016/j.meatsci.2010.02.001.
Marsh, J. J., and H. G. Lebherz. 1992. Fructose-bisphosphate aldolases: An evolutionary history. Trends Biochem. Sci. 17:110–113. https://doi.org/10.1016/0968-0004(92)90247-7.
Matsui, M., J. H. Fowler, and L. L. Walling. 2006. Leucine aminopeptidases: Diversity in structure and function. Biol. Chem. 387:1535–1544. https://doi.org/10.1515/BC.2006.191.
Matsushita-Morita, M., S. Tada, S. Suzuki, R. Hattori, J. Marui, I. Furukawa, Y. Yamagata, H. Amano, H. Ishida, M. Takeuchi, Y. Kashiwagi, and K. I. Kusumoto. 2011. Overexpression and characterization of an extracellular leucine aminopeptidase from Aspergillus oryzae. Curr. Microbiol. 62:557–564. https://doi.org/10.1007/s00284-010-9744-9.
Maurides, P. A., G. R. Akkaraju, and R. Jagus. 1989. Evaluation of protein phosphorylation state by a combination of vertical slab gel isoelectric focusing and immunoblotting. Anal. Biochem. 183:144–151. https://doi.org/10.1016/0003-2697(89)90182-6.
Mitra, B., R. Lametsch, I. Greco, and J. Ruiz-Carrascal. 2018. Advanced glycation end products, protein crosslinks and post translational modifications in pork subjected to different heat treatments. Meat Sci. 145:415–424. https://doi.org/10.1016/j.meatsci.2018.07.026.
Montowska, M., 2017. Using peptidomics to determine the authenticity of processed meat. In: M. Colgrave, editor, Proteomics in food science: From fark to fork. Academic Press, London, United Kingdom. p. 225–240. https://doi.org/10.1016/B978-0-12-804007-2.00014-X.
Montowska, M., M. R. Alexander, G. A. Tucker, and D. A. Barrett. 2014. Rapid detection of peptide markers for authentication purposes in raw and cooked meat using ambient liquid extraction surface analysis mass spectrometry. Anal. Chem. 86:10257–10265. https://doi.org/10.1021/ac502449w.
Montowska, M., M. R. Alexander, G. A. Tucker, and D. A. Barrett. 2015. Authentication of processed meat products by peptidomic analysis using rapid ambient mass spectrometry. Food Chem. 187:297–304. https://doi.org/10.1016/j.foodchem.2015.04.078.
Montowska, M., and E. Pospiech. 2013. Species-specific expression of various proteins in meat tissue: Proteomic analysis of raw and cooked meat and meat products made from beef, pork and selected poultry species. Food Chem. 136:1461–1469. https://doi.org/10.1016/j.foodchem.2012.09.072.
Nair, M. N., S. Li, C. M. Beach, G. Rentfrow, and S. P. Suman. 2018. Changes in the sarcoplasmic proteome of beef muscles with differential color stability during postmortem aging. Meat Muscle Biology. 2:1–17. https://doi.org/10.22175/mmb2017.07.0037.
Nair, M. N., S. P. Suman, M. K. Chatli, S. Li, P. Joseph, C. M. Beach, and G. Rentfrow. 2016. Proteome basis for intramuscular variation in color stability of beef semimembranosus. Meat Sci. 113:9–16. https://doi.org/10.1016/j.meatsci.2015.11.003.
NAMP. 2010. The Meat Buyers Guide. North Am. Meat Processors Assoc., Hoboken, NJ.
Nishimura, T., Y. Kato, A. Okitani, and H. Kato. 1991. Purification and properties of aminopeptidase C from chicken skeletal muscle. Agr. Biol. Chem. Tokyo. 55:1771–1778. https://doi.org/10.1080/00021369.1991.10870868.
Perng, M. D., L. Cairns, P. van den IJssel, A. Prescott, A. M. Hutcheson, and R. A. Quinlan. 1999. Intermediate filament interactions can be altered by HSP27 and alphaB-crystallin. J. Cell Sci. 112:2099–2112.
Pivovarova, A. V., V. V. Mikhailova, I. S. Chernik, N. A. Chebotareva, D. I. Levitsky, and N. B. Gusev. 2005. Effects of small heat shock proteins on the thermal denaturation and aggregation of F-actin. Biochem. Bioph. Res. Co. 331:1548–1553. https://doi.org/10.1016/j.bbrc.2005.04.077.
Polati, R., M. Menini, E. Robotti, R. Millioni, E. Marengo, E. Novelli, S. Balzan, and D. Cecconi. 2012. Proteomic changes involved in tenderization of bovine Longissimus dorsi muscle during prolonged ageing. Food Chem. 135:2052–2069. https://doi.org/10.1016/j.foodchem.2012.06.093.
Ponce, E., R. Linforth, M. Hall, I. Guerrero, and A. J. Taylor. 1994. Stability of haem pigments in model systems and cooked meat. Meat Sci. 38:141–151. dio:10.1016/0309-1740(94)90103-1.
Promeyrat, A., T. Sayd, E. Laville, C. Chambon, B. Lebret, and P. Gatellier. 2011. Early post-mortem sarcoplasmic proteome of porcine muscle related to protein oxidation. Food Chem. 127:1097–104. https://doi.org/10.1016/j.foodchem.2011.01.108.
Renerre, M., F. Dumont, and F. Gatellier. 1996. Antioxidant enzyme activities in beef in relation to oxidation of lipid and myoglobin. Meat Sci. 43:111–121. https://doi.org/10.1016/0309-1740(96)84583-9.
Rincon, A. M., R. K. Singh, and A. M. Stelzleni. 2015. Effects of endpoint temperature and thickness on quality of whole muscle non-intact steaks cooked in a radio frequency oven. LWT-Food Sci. Technol. 64:1323–1328. https://doi.org/10.1016/j.lwt.2015.07.017.
Roche, M., P. Rondeau, N. R. Singh, E. Tarnus, and E. Bourdon. 2008. The antioxidant properties of serum albumin. FEBS Lett. 582:1783–1787. https://doi.org/10.1016/j.febslet.2008.04.057.
Sarah, S. A., W. N. Faradalila, M. S. Salwani, I. Amin, S. A. Karsani, and A. Q. Sazili. 2016. LC-QTOF-MS identification of porcine-specific peptide in heat treated pork identifies candidate markers for meat species determination. Food Chem. 199:157–164. https://doi.org/10.1016/j.foodchem.2015.11.121.
Sarah, S. A., S. A. Karsani, I. Amina, N. F. K. Mokhtar, and A. Q. Sazili. 2014. A proteomic based assessment on changes in myofibrillar proteins of goat longissimus muscle as affected by heat treatments. J. Anim. Plant Sci. 24:406–412.
Sayd, T., C. Chambon, and V. Sante-Lhoutellier. 2016. Quantification of peptides released during in vitro digestion of cooked meat. Food Chem. 197:1311–1323. https://doi.org/10.1016/j.foodchem.2015.11.020.
Searcy, G. K., D. Senter, and R. L. Wilson. 1995. Glutamic-oxaloacetic transaminase activity: A potential end-point-temperature indicator for imported cooked beef. J. Food Protect. 58:686–688. https://doi.org/10.4315/0362-028X-58.6.686.
Suman, S. P., and P. Joseph. 2013. Myoglobin chemistry and meat color. Annu. Rev. Food Sci. T. 4:79–99. https://doi.org/10.1146/annurev-food-030212-182623.
Suman, S. P., M. C. Hunt, M. N. Nair, and G. Rentfrow. 2014. Improving beef color stability: Practical strategies and underlying mechanisms. Meat Sci. 98:490–504. https://doi.org/10.1016/j.meatsci.2014.06.032.
Suman, S. P., R. A. Mancini, R. Ramanathan, and M. R. Konda. 2009. Effect of lactate-enhancement, modified atmosphere packaging, and muscle source on the internal cooked colour of beef steaks. Meat Sci. 81:664–670. https://doi.org/10.1016/j.meatsci.2008.11.007.
Suman, S. P., R. A. Mancini, R. Ramanathan, and M. R. Konda. 2010. Modified atmosphere packaging influences premature browning in beef longissimus lumborum steaks. Fleischwirtschaft. 3:54–55.
Suman, S. P., M. N. Nair, P. Joseph, and M. C. Hunt. 2016. Factors influencing internal color of cooked meats. Meat Sci. 120:133–144. https://doi.org/10.1016/j.meatsci.2016.04.006.
Szklarczyk, D., A. Franceschini, S. Wyder, K. Forslund, D. Heller, J. Huerta-Cepas, M. Simonovic, A. Roth, A. Santos, K. P. Tsafou, M. Kuhn, P. Bork, L. J. Jensen, and C. Von Mering. 2015. STRING v10: Protein-protein interaction networks, integrated over the tree of life. Nucleic Acids Res. 43:D447–D452. https://doi.org/10.1093/nar/gku1003.
Taylor, A. J., R. Linforth, O. Weir, T. Hutton, and B. Green. 1993. Potential of electrospray mass spectrometry for meat pigment identification. Meat Sci. 33:75–83. https://doi.org/10.1016/0309-1740(93)90095-Y.
Tian, X., W. Wu, Q. Yu, M. Hou, F. Jia, X. Li, and R. Dai. 2016. Quality and proteome changes of beef M. longissimus dorsi cooked using water bath and ohmic heating process. Innov. Food Sci. Emerg. 34:259–266. https://doi.org/10.1016/j.ifset.2016.02.013.
Toldra, F., M. C. Aristoy, C. Part, C. Cervero, E. Rico, M. J. Motilva, and J. Flores. 1992. Muscle and adipose tissue aminopeptidase activities in raw and dry-cured ham. J. Food Sci. 57:816–818. https://doi.org/10.1111/j.1365-2621.1992.tb14301.x.
Tornberg, E. 2005. Effects of heat on meat proteins: Implications on structure and quality of meat products. Meat Sci. 70:493–508. https://doi.org/10.1016/j.meatsci.2004.11.021.
Van Laack, R. L. J. M., B. W. Berry, and M. B. Solomon. 1996. Variations in internal color of cooked beef patties. J. Food Sci. 61:410–414. https://doi.org/10.1111/j.1365-2621.1996.tb14205.x.
Watson, A. D., Y. Gunning, N. M. Rigby, M. Philo, and E. K. Kemsley. 2015. Meat authentication via multiple reaction monitoring mass spectrometry of myoglobin peptides. Anal. Chem. 87:10315–10322. https://doi.org/10.1021/acs.analchem.5b02318.
Wen, S., G. Zhou, L. Li, X. Xu, X. Yu, Y. Bai, and C. Li. 2015. Effect of cooking on in vitro digestion of pork proteins: A peptidomic perspective. J. Agr. Food Chem. 63:250–261. https://doi.org/10.1021/jf505323g.
Wood, Z. A., E. Schroder, J. R. Harris, and L. B. Poole. 2003. Structure, mechanism and regulation of peroxiredoxins. Trends Biochem. Sci. 28:32–40. https://doi.org/10.1016/S0968-0004(02)00003-8.
Wu, W., X. G. Gao, Y. Dai, Y. Fu, X. M. Li, and R. T. Dai. 2015b. Post-mortem changes in sarcoplasmic proteome and its relationship to meat color traits in M. semitendinosus of Chinese Luxi yellow cattle. Food Res. Int. 72:98–105. https://doi.org/10.1016/j.foodres.2015.03.030.
Wu, W., Q. Q. Yua, Y. Fuc, X. J. Tian, F. Jia, X. M. Li, and R. T. Dai. 2015a. Towards muscle-specific meat color stability of Chinese Luxi yellow cattle: A proteomic insight into post-mortem storage. J. Proteomics. 147:108–118. https://doi.org/10.1016/j.jprot.2015.10.027.
Yan, Y. B., Q. Wang, H. W. He, and H. M. Zhou. 2004. Protein thermal aggregation involves distinct regions: Sequential events in the heat-induced unfolding and aggregation of hemoglobin. Biophys. J. 86:1682–1690. https://doi.org/10.1016/S0006-3495(04)74237-X.
Yancey, J. W. S., J. K. Apple, and M. D. Wharton. 2016. Cookery method and endpoint temperature can affect the Warner-Bratzler shear force, cooking loss, and internal cooked color of beef semimembranosus and infraspinatus steaks. J. Anim. Sci. 94:4434–4446. https://doi.org/10.2527/jas.2016-0651.
Yancey, J. W. S., M. D. Wharton, and J. K. Apple. 2011. Cookery method and end-point temperature can affect the Warner-Bratzler shear force, cooking loss, and internal cooked color of beef longissimus steaks. Meat Sci. 88:1–7. https://doi.org/10.1016/j.meatsci.2010.11.020.
Yu, T. Y., J. D. Morton, S. Clerens, and J. M. Dyer. 2015. Proteomic investigation of protein profile changes and amino acid residue level modification in cooked lamb meat: The effect of boiling. J. Agr. Food Chem. 63:9112–9123. https://doi.org/10.1021/acs.jafc.5b03324.
Yu, T. Y., J. D. Morton, S. Clerens, and J. M. Dyer. 2016. Proteomic investigation of protein profile changes and amino acid residue-level modification in cooked lamb longissimus thoracis et lumborum: The effect of roasting. Meat Sci. 119:80–88. https://doi.org/10.1016/j.meatsci.2016.04.024.
Yu, T. Y., J. D. Morton, S. Clerens, and J. M. Dyer. 2017. Cooking-induced protein modifications in meat. Compr. Rev. Food Sci. F. 17:141–159. https://doi.org/10.1111/1541-4337.12243.
Zhai, C., S. P. Suman, M. N. Nair, S. Li, X. Luo, C. M. Beach, B. N. Harsh, D. D. Boler, A. C. Dilger, and D. W. Shike. 2018. Supranutritional supplementation of vitamin E influences mitochondrial proteome profile of post-mortem longissimus lumborum from feedlot heifers. S. Afr. J. Anim. Sci. 48:1140–1147. https://doi.org/10.4314/sajas.v48i6.18.
Zhang, Y. L., J. M. Zhou, and C. L. Tsou. 1993. Inactivation precedes conformation change during thermal denaturation of adenylate kinase. BBA-Protein Struct. M. 1164:61–67. https://doi.org/10.1016/0167-4838(93)90112-5.
Zhu, K., J. Zhao, D. M. Lubman, F. R. Miller, and T. J. Barder. 2005. Protein pI shifts due to posttranslational modifications in the separation and characterization of proteins. Anal. Chem. 77:2745–2755. https://doi.org/10.1021/ac048494w.